Scholarship opportunities
UQ Graduate School Scholarship (UQGSS)
The UQ Graduate School Scholarship is the most common scholarship applied for when planning to undertake a Phd. Most PhD's at UQ are funded via the UQ Graduate School Scholarship. For more information on eligibility and how to apply, please visit the UQ Graduate School.

Earmarked Scholarships
Earmarked scholarships are offered for research projects aligned with recently awarded grants from major Australian Government research funding bodies, including the Australian Research Council, the National Health and Medical Research Council and the Medical Research Future Fund.
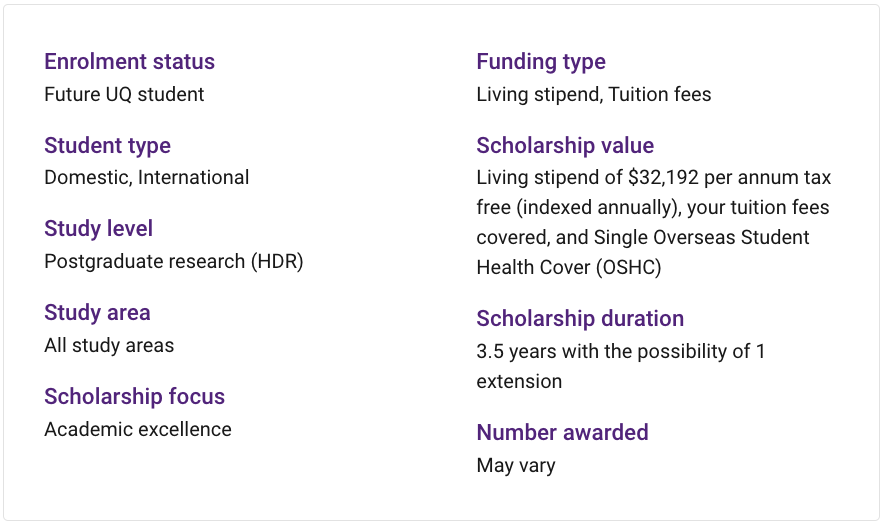
Earmarked scholarships offer PhD students the opportunity to contribute to large projects of national significance and work with leading researchers. The scholarship provides a living stipend of $32,192 per annum (2023 rate, indexed annually), tuition fees, and Overseas Student Health Cover (OSHC). You can apply anytime - there is no closing date.
IMB Earmarked Scholarships are added to the PhD Projects list as they become available. For more information on Earmarked Scholarships and how to apply, please visit the UQ Graduate School website.
Simon Axelsen Anderson PhD scholarships
UQ's Institute for Molecular Bioscience (IMB) is launching this new PhD scholarship funded through distributions from the Simon Anders Axelsen Memorial Fund.
Interested candidates will submit Expressions of Interest directly to the relevant Principal Advisor, after which IMB will internally review and rank applicants and nominate the top candidate for admission and scholarship consideration.
For more specific scholarship information, please view the scholarships listing.
Project Title: Harnessing Machine Learning to Design Retromer-Stabilising Therapeutics for Neurodegenerative Diseases
Short project description:
The disruption of proteostasis, the regulation of protein dynamics within neural cells, is a central mechanism in neurodegenerative diseases. This disruption results in the accumulation of toxic proteins, such as amyloid beta (Aβ) and phosphorylated tau. The endolysosomal system largely controls proteostasis, with the Retromer complex acting as a critical regulator. In Alzheimer's disease, Retromer is significantly downregulated in the hippocampus. While stabilising Retromer with molecular chaperones has demonstrated potential in increasing Retromer levels and decreasing Aβ, current molecules lack clinical viability.
This project aims to develop novel, neuroprotective therapeutic candidates by combining expertise in membrane trafficking with advanced machine learning techniques, specifically AlphaFold2 and RFdiffusion. By designing novel protein backbones and small macrocyclic molecules targeting specific regions of Retromer, we aim to address a fundamental molecular pathology common to neurodegenerative diseases. This will be achieved through the following specific aims:
Design and Validation of Chaperones: Design a comprehensive suite of protein and peptide chaperones utilizing machine learning, and rigorously evaluate their efficacy in stabilising the Retromer complex both in vitro and in cellular models.
Structural Confirmation and Optimisation: Conduct detailed structural studies to verify the accuracy of the computational designs and perform iterative fine-tuning of the chaperones based on structure-activity relationships.
Phenotypic Efficacy in Cellular Models: Assess the capacity of the optimised chaperones to significantly reduce established phenotypic markers of neurodegeneration within relevant cellular models.
Principal Advisor name: Michael Healy
Co-supervisor: Professor David Ascher from SCMB (https://scmb.uq.edu.au/profile/8654/david-ascher) has said that he would be happy to co-supervise a student.
Project title: Unlocking Genetic Insights into Alzheimer’s Disease Through AI-Predicted Hippocampal Volume
Project Description: Alzheimer’s disease (AD) is a devastating neurodegenerative disorder where hippocampal atrophy is a critical biomarker, directly linked to memory loss and cognitive decline. Understanding the genetic architecture of hippocampal volume is essential for uncovering biological mechanisms underlying AD and for identifying novel therapeutic targets. However, genetic discovery is limited by the scarcity of MRI data in large cohorts—only ~100,000 of the UK Biobank’s ~500,000 participants have neuroimaging, leaving a vast untapped resource for genetic research.
This project will harness AI to predict hippocampal volume, enabling groundbreaking genetic analyses. By training machine/deep learning models on UK Biobank data (demographics, clinical, lifestyle, cognitive, and biomarker variables), we will generate AI-derived hippocampal volumes for participants without MRI scans. This approach will expand the effective sample size for genetic studies, increasing statistical power to detect novel AD-associated loci.
Key aims:
· Train and validate AI models to predict hippocampal volume using non-imaging data.
· Apply the best-performing model to ~400,000 non-imaged UK Biobank participants.: · Perform GWAS on both observed and AI-predicted hippocampal volumes to identify new genetic variantslinked to hippocampal volume.
· Validate findings on external AD cohorts.
Principal Advisor: Prof Allan McRae (IMB, UQ)
Co-Supervisor: A/Prof Baptiste Couvy-Duchesne (QIMR)
Project title: Targeting Novel Ion Channel Pathways in Pain Management
Project description: Are you ready to work at the cutting edge of pain research and help change how the world treats pain? This PhD project invites an ambitious student to join a leading team exploring how targeting ion channels, the molecular gates that control how nerve cells sense and transmit pain, may lead to better pain management. When these channels malfunction, such as in inflammatory pain caused by gout or inflammatory bowel disease, patients can experience relentless, life-altering discomfort. Understanding these pathways is essential for developing safer and more effective treatments. A deeper understanding of these mechanisms is critical for the development of safer, more precise, and more effective treatments.
In this project, you will investigate how blocking specific ion channels alters pain perception and the activation of immune and neuronal cells. You will use advanced tools such as high‑resolution imaging, electrophysiology, and modern cell‑based models to uncover how these channels drive pain. You will also work with naturally occurring bioactive molecules, including venom‑derived peptides, which act as highly selective probes to pinpoint key channels involved in pain. These molecules may help lead to next‑generation analgesics with fewer side effects.
You will join a vibrant research environment equipped with state‑of‑the‑art technology and supported by experts in behavioural neuroscience, neuroinflammation, sensory neuropharmacology, and toxin‑based analgesic discovery. This is a unique opportunity to gain high‑value skills, contribute to meaningful scientific advances, and position yourself at the forefront of innovative pain research.
If you want to be part of a project that combines scientific discovery, real‑world impact, and exciting career potential, this is your opportunity. Step into a field where your work could shape the future of pain management.
Principal Advisor: Dr Svetlana Shatunova, Institute for Molecular Bioscience (IMB), The University of Queensland
Co-supervisor: Dr David Klyne, School of Health and Rehabilitation Sciences, The University of Queensland
Project Title: Decoding Pain: Linking Neural Circuits to Behaviour to Transform Treatment
Project Description: Chronic and severe pain remains one of the greatest unmet challenges in medicine. Despite decades of research, many treatments fail because we still do not fully understand how the nervous system transforms a physical stimulus into the experience of pain. In most laboratory studies, pain is measured by observing behaviour in rodents, such as a paw withdrawal, but we rarely know which neural circuits are driving that response. Bridging this gap is essential if we want to design better therapies.
This exciting project will take a systems-level approach to decoding pain. Using precisely controlled thermal and mechanical stimuli, we will map which nerve cells are activated across the sensory nerves, spinal cord and brain. By combining advanced neural activity tagging, high-resolution imaging, computational mapping and AI-assisted behavioural analysis, we will build integrated maps that connect stimulus intensity to neural activation patterns and behavioural outcomes.
Importantly, we will apply this framework to clinically relevant pain conditions, including chemotherapy-induced neuropathy, to determine whether hypersensitivity reflects stronger activation of normal circuits or the recruitment of entirely new neural pathways.
Students joining this project will gain hands-on training in cutting-edge neuroscience techniques spanning circuit mapping, transcriptomics, behavioural phenotyping and computational analysis. This is a unique opportunity to work at the interface of neuroscience, immunology and translational medicine, contributing to discoveries that could reshape how pain is understood and treated.
Principal Advisor: Dr Hana Starobova, Institute for Molecular Bioscience (IMB), The University of Queensland
Co-supervisor: Prof. Paul Hodges, School of Health and Rehabilitation Sciences, The University of Queensland
Project title: Investigating the role of epidermal growth factor receptor (EGFR) signalling in pain
Project description:
Epidermal growth factor receptors (EGFRs; ERBB1–4) are receptor tyrosine kinases that function as homo- or heterodimers and are best known for their roles in development and cancer biology. Recent clinical observations that cancer patients receiving EGFR inhibitors report reduced pain suggest that EGFR signalling may also contribute to chronic pain. Supporting this idea, the endogenous EGFR ligand epiregulin has been shown to potentiate nociceptive signalling. Furthermore, we have discovered that some stinging ants produce novel EGFR ligands in their venoms that act on sensory neurons to enhance the pain-causing effects of other venom components. These findings lead us to hypothesise that a specific ERBB receptor expressed in adult mammalian sensory neurons mediates the potentiation of nociceptive signalling, and that inhibition of this receptor could suppress pain signalling.
This project will identify and characterise the ERBB receptor responsible, including its subtype (and possible splice variants), cell-specific expression, signalling pathways,and ligand specificity. We will also determine how nerve injury affects its expression and evaluate its potential as a therapeutic target for chronic pain. To achieve this, we will generate and characterise a toolkit of novel EGFR ligands from ant venoms and assess their potency and selectivity. Using endogenous ligands, venom-derived ligands, and clinically available EGFR inhibitors, we will examine modulation of pain signalling in vitro and in vivo. Transcriptomics, immunohistochemistry, and pharmacological approaches will be used to identify the receptor subtype(s) involved. By defining the role of EGFR signalling in sensory neurons, this project aims to uncover a novel therapeutic target that could enable the development of new treatments for chronic pain.
Principal Advisor: Samuel Robinson (IMB)
Co-supervisor: Irina Vetter (School of Pharmacy/IMB)
Project Title: Venoms to Drugs: Peptide analgesics for the treatment of severe chronic pain.
Project Description: Severe chronic pain remains a major unmet medical need, with current analgesics having limited efficacy or unacceptable side‑effects. This PhD project addresses these shortcomings by developing next-generation bispecific peptide analgesics that act through non-opioid mechanisms, blocking ion channel activity in pain-sensing neurons.
We are particularly interested in targeting voltage-gated sodium (NaV) channel subtypes that play critical roles in peripheral-to-central pain signalling. Taking advantage of the vast source of bioactive peptides in venoms that interact with ion channels through different mechanisms, we will design and synthesise dual-site peptide ligands combining gating-modifiers, pore blockers, and interacting-protein binders to stabilise analgesic NaV conformations through synergistic dual-site mechanisms. Using multidisciplinary approaches spanning peptide and medicinal chemistry, structure–activity optimisation, electrophysiology, and molecular pharmacology, we aim to break new ground in ion channel targeting and the development of next-generation, non-addictive precision analgesics.
The project will deliver: (i) powerful new tools for studying ion-channel biology in the context of severe pain; (ii) fundamental insights into dual-site ligand–channel interactions that underpin durable analgesia; and (iii) broadly applicable design strategies for long-acting, selective ion-channel modulators for the development of next-generation pain therapeutics.
Techniques that will be learned:
- Peptide and Medicinal Chemistry
- Drug Design and Development
- Ion channel pharmacology
- Cell culture and bioassays
- Confocal Microscopy
- Analytical techniques (HPLC, LCMS, NMR, CD)
- Computational modelling (AlphaFold, ligand-receptor modelling)
Principal Advisor: A/Prof. Markus Muttenthaler
Co-Advisor: A/Prof. Lachlan Rash
Project Title: Targeting the Oxytocin Receptor for Cancer Diagnosis and Treatment
Project Description: Breast cancer and prostate cancer are the most commonly diagnosed cancers in women and men, respectively, and remain the leading causes of cancer burden worldwide. Effective treatment options are limited for late-stage cancers, triple-negative breast cancer and cancers that develop treatment resistance, resulting in poor patient outcomes.
The oxytocin receptor (OTR) is a G protein-coupled receptor best known for roles in social bonding and reproduction, including parent–child/partnership bonding, sexual arousal, parturition and breastfeeding. OTR is also involved in cancer development, with elevated OTR expression in late-stage breast and prostate cancer cells/tumours and associations with increased aggressiveness and metastatic behaviour. OTR represents therefore an attractive yet underexplored target for tumour visualisation/detection procedures and treatment to improve cancer management.
The Muttenthaler research group is world-leading in OTR research and has recently developed OTR-specific near-infrared and PET/CT tracers to improve cancer diagnosis and treatment. This project focuses on characterising and advancing these tracers further for applications such as monitoring treatment response, detecting primary tumours and recurrences, and guiding surgeons during tumour removal.
Overall, this project will advance our understanding of OTR’s role and therapeutic scope in cancer biology. Translation of tumour-specific, highly sensitive tracers developed in this project would have a substantial impact on breast and prostate cancer management, reduce healthcare costs, and, most importantly, reduce mortality through earlier, more reliable detection, improved response monitoring and more accurate surgical resection.
Techniques that will be learned:
- Peptide and Medicinal Chemistry
- Theranostic Design and Development
- Neuropeptide and GPCR pharmacology
- Molecular and Cancer Biology
- Confocal Microscopy and other imaging techniques (PET/CT, NIR)
- Cell culture and bioassays
- Analytical techniques (HPLC, LCMS, NMR, CD)
Principal Advisor: A/Prof. Markus Muttenthaler
Co-Advisor: A/Prof. Karine Mardon
Project Title: Venom-derived peptides as next-generation therapeutics for pain
Project Description: Chronic pain remains a major unmet clinical challenge, and we urgently require new drugs that can provide relief without side effects. The Vetter group at the IMB uses animal venom-derived peptides as new therapeutic leads for the development of innovative analgesic. Many venom-derived peptides target ion channels that control neuronal excitability, including voltage-gated sodium (NaV), potassium (Kv) and calcium channels that play central roles in pain signalling. These peptides are often extraordinarily potent and highly selective, allowing precise modulation of specific molecular targets with minimal off-target effects.
The project will combine cutting-edge interdisciplinary approaches spanning molecular pharmacology, chemical biology, neuroscience and drug discovery.
The successful candidate will contribute to development of new experimental platforms to identify and characterise peptide modulators of ion channels involved in pain signalling. The project will integrate advanced cell-based and in vivo approaches to understand how peptide toxins interact with sensory neuron targets and how these interactions can be harnessed to develop novel analgesic strategies.
This project offers training in a broad range of state-of-the-art techniques, including:
- molecular and cellular assays to investigate ion channel function in mammalian cells
- isolation and culture of primary sensory neurons
- peptide synthesis and rational peptide engineering
- high-throughput and high-content imaging approaches
- manual and automated patch-clamp electrophysiology
- pharmacological characterisation of ion channel modulators
Research environment
Prof Vetter is an internationally recognised leader in ion channel pharmacology and venom peptide research and was awarded the University of Queensland Award for Excellence in Research Higher Degree Supervision in 2019 as the youngest recipient of this award. The research group provides a supportive and stimulating training environment comprising PhD, Masters, Honours and undergraduate students together with a team of experienced postdoctoral researchers who contribute mentorship and technical expertise.
Principal Advisor: Prof Irina Vetter
Co-Advisor: Prof Mehdi Mobli (AIBN)
Project Title: Lipase–Host Lipid Interactions in Chronic Salmonella Infection and Gallbladder Carcinogenesis
Project Description: Chronic infection with Salmonella enterica is the major risk factor for gallbladder carcinoma, a highly aggressive malignancy with poor clinical outcomes. In endemic regions, long-term biliary carriage of Salmonella is strongly associated with gallbladder inflammation, altered lipid composition, and malignant transformation, yet the molecular mechanisms linking bacterial persistence to cancer development remain poorly defined.
This PhD project will investigate the role of Salmonella-derived lipases in modulating host lipid biology during chronic infection and how these interactions contribute to gallbladder carcinogenesis. The central hypothesis is that bacterial lipase activity alters host lipid pools and membrane dynamics within the gallbladder environment, promoting chronic inflammation, epithelial dysregulation, and tumour‑promoting signalling pathways.
The successful candidate will use a multidisciplinary approach combining bacterial genetics, infection biology, lipid biochemistry, and host–pathogen interaction models. Experimental strategies may include bacterial mutant analysis, infection of epithelial and macrophage models, lipid profiling, and mechanistic studies of host signalling and stress responses. There will be opportunities to tailor aspects of the project to the candidate’s interests, including cancer biology, microbial metabolism, or host lipid regulation.
This project offers comprehensive training in molecular microbiology, cell biology, and advanced analytical techniques, as well as experience in experimental design, data analysis, and scientific communication. The work has strong translational relevance, with implications for understanding infection‑driven cancers and identifying novel therapeutic or preventative targets.
Ideal candidates will have a background in microbiology, biochemistry, molecular biology, or a related discipline, with a strong interest in host–pathogen interactions and infection‑associated disease.
Principal Advisor: Professor Ian Henderson
